Back
Sensitive and accurate analyses of free 3-nitrotyrosine in exhaled breath condensate by LC-MS/MS.
T. Göen, A. Müller-Lux, P. Dewes, A. Musiol, T. Kraus
J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2005 Nov;826(1-2):261-6.
PubMed: 16143570
Abstract
The quantitative determination of 3-nitro-l-tyrosine, a biological marker for inflammatory processes, in exhaled breath condensate (EBC) is described. The clean-up and preconcentration was performed by solid phase extraction (SPE). After liquid chromatography the specific detection was performed by tandem mass spectrometry using electron spray ionisation and selected reaction monitoring (SRM). 13C9-3-nitrotyrosine was used as an internal standard. For reliability, tests for the precision of the method, the losses during preparation, a test for nitrating artifacts and the comparibility of calibrants in EBC and buffer solution were performed. The calibration of the method was linear over a range of 10-500 pg/mL. The within-run coefficients of variation (CV) of the samples were found to be 8.4% at 25 pg/mL and 8.3% at 250 pg/mL. The day-to-day CV was found to be 11.2%. The limit of quantification was 3.9 pg/mL. The losses during preparation were 15%. The discrepancy between the calibration with EBC and buffer solution was below 10%. No artificial production of 3-nitrotyrosine was observed during the procedure. The application of the method on the EBC samples of healthy smokers (N=10) and non-smokers (N=10) showed no difference between the two groups. The concentration of 3-nitrotyrosine ranged between the limit of quantification and 184 pg/mL and was distinctly lower than data detected by an immunoassay procedure. The procedure was proven to be accurate, sensitive and in contrast to GC methods less elaborate and is recommended for the determination of 3-nitrotyrosine in exhaled breath condensate.
Associated compounds:
|
Compound Name
with link to compound page |
Structure | Number of references |
|---|---|---|
| 3-Nitrotyrosine | 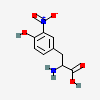 |
15 |
